Publications
For a full list see below. I'll make my best effort to keep this list updated, but please also check Google Scholar , ORCID , and ResearchGate.
Group Highlights
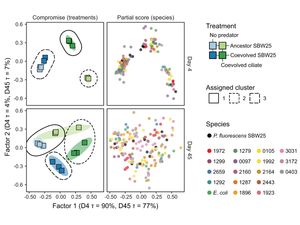
Predator/prey coevolution and microbial community-wide gene expression
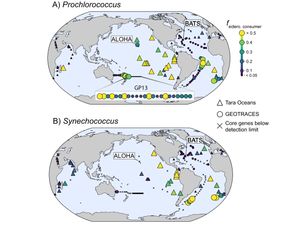
Iron scavenging by marine picocyanobacteria
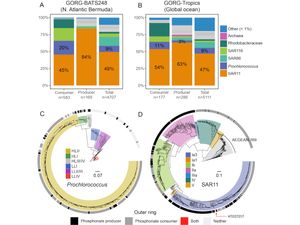
Microbial sources of organophosphonates to the marine P cycle
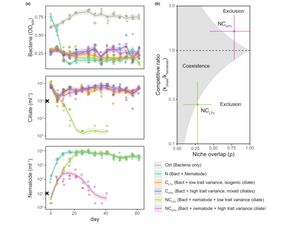
Intraspecific trait variability and species coexistence
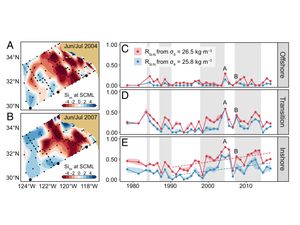
Phytoplankton iron limitation at marine deep chlorophyll maximum (DCM) layers
All publications
‡ indicates equal contributions
◇ indicates corresponding author(s)
Boldface indicates SL Hogle and Ph.D./M.Sc. students formally supervised
[17] Hogle SL◇, Ruusulehto L, Cairns J, Hultman J, Hiltunen T◇. Localized coevolution between microbial predator and prey alters community-wide gene expression and ecosystem function. ISME Journal, 17, 514-524 (2023). doi: 10.1038/s41396-023-01361-9 Open Access
[16] Hackl T◇‡, Laurenceau ‡, Ankenbrand M‡, Bliem C, Cariani Z, Thomas E, Dooley K, Arellano A, Hogle SL, Berube P, Leventhal G, Luo E, Eppley J, Zayed A, Beaulaurier J, Stepanauskas R, Sullivan M, DeLong E, Biller S, Chisholm SW◇. Novel integrative elements and genomic plasticity in ocean ecosystems. Cell, 186, 47-62 (2023). doi: 10.1016/j.cell.2022.12.006
[15] Hogle SL◇, Hackl T, Bundy R, Park J, Satinsky B, Hiltunen T, Biller S, Berube P, Chisholm SW◇. Siderophores as an iron source for picocyanobacteria in deep chlorophyll maximum layers of the oligotrophic ocean. ISME Journal, 16, 1636-1646 (2022). doi: 10.1038/s41396-022-01215-w Open Access
[14] Acker M‡, Hogle SL‡, Berube P, Hackl T, Stepanauskas R, Chisholm SW, Repeta DJ◇. Phosphonate production by marine microbes: exploring new sources and potential function. Proc. Natl. Acad. Sci. USA, 119, e2113386119 (2022). doi: 10.1073/pnas.2113386119 Open Access
[13] Hogle SL◇‡, Hepolehto I‡, Ruokolainen L, Cairns J, Hiltunen T◇. Effects of phenotypic variation on consumer coexistence and prey community structure. Ecol. Lett, 25, 307-319 (2022). doi: 10.1111/ele.13924 Open Access
[12] Becker J◇, Hogle SL, Rosendo K, Chisholm SW◇. Co-culture and biogeography of Prochlorococcus and SAR11. ISME Journal, 13, 1506-1519 (2019). doi: 10.1038/s41396-019-0365-4 Open Access
[11] Hogle SL◇, Dupont C, Hopkinson B, King A, Buck K, Roe K, Stuart R, Allen A, Mann E, Johnson Z, Barbeau K◇. Pervasive iron limitation at subsurface chlorophyll maxima of the California Current. Proc. Natl. Acad. Sci. USA, 115, 13300-13305 (2018). doi: 10.1073/pnas.1813192115 Open Access
[10] Berube P◇, Biller S, Hackl T, Hogle SL, Satinsky B, Becker J, Braakman R, Collins S, Kelly L, Berta-Thompson J, Coe A, Bergauer K, Bouman H, Browning T, De Corte D, Hassler C, Hulata Y, Jacquot J, Maas E, Reinthaler T, Sintes E, Yokokawa T, Lindell D, Stepanauskas R, Chisholm SW◇. Single cell genomes of Prochlorococcus, Synechococcus, and sympatric microbes from diverse marine environments. Scientific Data, 5, 180154 (2018). doi: 10.1038/sdata.2018.154 Open Access
[9] Biller S◇, Berube P, Dooley K, Williams M, Satinsky B, Hackl T, Hogle SL, Coe A, Bergauer K, Bouman H, Browning T, De Corte D, Hassler C, Hulston D, Jacquot J, Maas EW, Reinthaler T, Sintes E, Yokokawa T, Chisholm SW◇. Marine microbial metagenomes sampled across space and time. Scientific Data, 5, 180176 (2018). doi: 10.1038/sdata.2018.176 Open Access
[8] Hogle SL◇, Brahamsha B, Barbeau K. Direct Heme Uptake by Phytoplankton-Associated Roseobacter Bacteria. mSystems, 2, e00124-16 (2017). doi: 10.1128/mSystems.00124-16 Open Access
[7] Hogle SL◇, Bundy R, Blanton J, Allen E, Barbeau K. Copiotrophic marine bacteria are associated with strong iron-binding ligand production during phytoplankton blooms. Limnol. Oceanogr. Lett., 1, 36-43 (2016). doi: 10.1002/lol2.10026 Open Access
[6] Hogle SL◇, Thrash, JC, Dupont C, Barbeau K. Trace metal acquisition by marine heterotrophic bacterioplankton with contrasting trophic strategies. Appl. Environ. Microbiol., 82, 1613-1624 (2016). doi: 10.1128/AEM.03128-15 Open Access
[5] Dupont C, McCrow J, Valas R, Moustafa A, Walworth N, Goodenough U, Roth R, Hogle SL, Bai J, Johnson Z, Mann E, Palenik B, Barbeau K, Venter JC, Allen A◇. Genomes and gene expression across light and productivity gradients in eastern subtropical Pacific microbial communities. ISME Journal, 9, 1076-1092 (2015). doi: 10.1038/ismej.2014.198 Open Access
[4] Hogle SL◇, Barbeau K, Gledhill M. Heme in the marine environment: from cells to the iron cycle. Metallomics, 6, 1107-1120 (2014). doi: 10.1039/c4mt00031e
[3] Kharbush J◇, Ugalde J, Hogle SL, Allen E, Aluwihare L. Composite bacterial hopanoids and their microbial producers across oxygen gradients in the water column of the California Current. Appl. Environ. Microbiol., 79, 7491-7501 (2013). doi: 10.1128/AEM.02367-13
[2] Roe K◇, Hogle SL, Barbeau K. Utilization of heme as an iron source by marine alphaproteobacteria in the roseobacter clade. Appl. Environ. Microbiol., 79, 5753-5762 (2013). doi: 10.1128/AEM.01562-13
[1] Abbriano R, Carranza M, Hogle SL, Levin R, Netburn A, Seto K, Snyder S, Franks P◇. Deepwater Horizon Oil Spill: A Review of the Planktonic Response. Oceanography, 24, 294-301 (2011). doi: 10.5670/oceanog.2011.80 Open Access